News
eMurmur Digital Stethoscope Platform Datasheet
ri-sonic® PCP Telemedicine Stethoscopes w/eMurmur Datasheet
Joint Press Release Let's Talk Interactive / MobilDrTech
PCP-USB/PCP-SSP Telemedicine Stethoscope Datasheet
RCS-100 General Examination Camera Datasheet
RCS-100 Comparison to Horus Scope Series 2
Let's Talk Interactive Telemedicine Software Platform
RVS-100 Vital Signs Monitor Datasheet
RVS-100 Vital Signs Monitor Brochure
White Paper: Telemedicine Stethoscopes - Updated July, 2020
Comparison Charts - Telemedicine Stethoscopes - Updated July, 2020
QT Medical PCA 500 Portable, 12-Lead ECG System - Download
BrochurePress Release - MobilDrTech to Carry Portable Power System for Telemedicine
Pack-Power™ Portable Power System for Telemedicine On The Go
Why Select PCP-USB Telemedicine Stethoscope
MobilDrTech Partners with BodiMetrics to Bring RPM Home
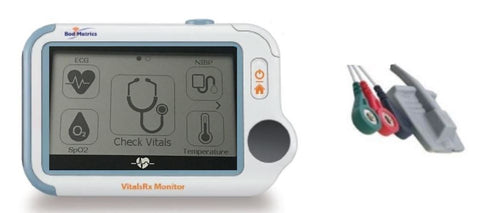
MobilDrTech partners with vitals device maker BodiMetrics™ to market FDA 510k approved VitalsRx Monitor with spot vitals and continuous ECG and SpO2 monitoring.
HOUSTON, TEXAS, USA, June 13, 2019 /EINPresswire.com/ -- Remote Physiologic Monitoring (RPM) and Chronic Care Management (CCM) are two rapidly growing areas of medicine. In early 2018, CMS "unbundled" the CPT code for Remote Patient Care, 99091. This separated qualifying services from existing telehealth and telemedicine reimbursement guidelines. This has opened the door for healthcare providers to be reimbursed for some RPM services including data collection and care coordination for patients, including those at home.
MobilDrTech, Inc. and BodiMetrics™ announce a strategic partnership to market professional and consumer grade collection and analysis of health vitals with pocket-sized devices to improve management of chronic conditions, enhance quality of fitness and wellness programs and optimize clinical trials and research.
VitalsRx Features and Metrics
• 20 Second ECG Waveform
• Continuous ECG to 24 Hrs - 24 Hour Holter Monitoring
• 20 Second SpO2 Capture
• Continuous SpO2 to 15 Hrs - Overnight Screening for Sleep Apnea
• Heart Rate & Variability
• 3 Second Thermometer
• Stress Relief Bio-Feedback
• Pedometer Steps
• Medication Reminders - Ideal for Pharmaceutical Research Projects
BodiMetrics™ Cloud is a powerful system that provides patients and clinicians secure and easy connections. Data can flow from one person to another seamlessly and freely under authorization. Both patients and clinicians benefit from the availability of data.
• Data can be synced to cloud automatically
• Data can be shared with clinician automatically
• Data can be stored in cloud over long term for health tracking
• Data is encrypted to protect user privacy
Capture Spot Vitals and Continuous ECG / SpO2
Connect Via Bluetooth to IOS or Android Devices with Free App
VitalsRx Monitor and Included Accessories
About BodiMetrics
BodiMetrics is dedicated to empowering consumers with products, services and solutions that will allow them to take charge of their healthcare, lower overall cost and improve their well-being. Products are tested by independent third party labs to clinical standards as well, where indicated, approved by the FDA. http://www.bodimetrics.com
About MobilDrTech
MobilDrTech, Inc. is a full-service telemedicine systems and equipment provider. Founded in 2009 and based in Sugar Land, TX, MobilDrTech provides telemedicine services such as HIPAA compliant video conference platforms and functions as a Value-Added Reseller (VAR) for telemedicine equipment manufacturers including leading manufacturers of telemedicine stethoscopes, otoscopes, dermascopes, general examination cameras, telemedicine carts and vitals monitoring devices. http://www.mobildrtech.com
Robert Jenkins
MobilDrTech, Inc.
+1 281-340-2013
email us here
MobilDrTech Announces Collaboration with QT Medical for Release of New 12-Lead ECG
Sugar Land, TX, February 11, 2019
MobilDrTech, Inc. has entered into a partnership with award winning startup QT Medical to introduce the PCA 500, the world’s most compact, wireless, 12-lead ECG system for personal use. https://www.telemedicinesupply.com/collections/ecgs .
Last month at the Consumer Electronics Show (CES) in Las Vegas, QT Medical announced the launch of its Personal Cardiac Assistant (PCA 500), the only FDA-cleared, hospital quality, 12-lead electrocardiogram (ECG) for personal use. 
Several health trackers including Apple Watch 4 can record single lead ECGs and identify arrhythmias, including atrial fibrillation or AFib. Hospital grade 12-lead ECGs do much more and are used to diagnose a variety of cardiovascular conditions including arrhythmias, ischemic changes, location of changes (anterior, inferior), QT interval changes, etc.
Because special training is required for the operator to place the leads and conduct the ECG, 12-lead ECG diagnostics has historically been limited to clinical settings. QT Medical’s Personal Cardiac Assistant (PCA 500) is the first hospital grade, 12-lead ECG designed for use by the patient without trained medical personnel. 
PCA 500 is digital, wireless, and mobile friendly. It is smaller than a deck of cards, fits in the shirt pocket, and works with iOS and Android phones and tablets.
The system uses flexible strips to combine the electronic leads and to help position them around the chest area. Instructions meant to be easily understood by average people help to make sure that each of the twelve leads is placed in the correct spot. Once the system is ready and the leads are placed, the control unit is activated to make a recording.
In a clinical trial, it was found that over 95% of patients with no training, and no experience with ECGs, could do a full 12-lead ECG test after reading simple instructions.
This revolutionary system will enable patients with heart disease to do timely, hospital quality, 12-lead ECG tests at their convenience and in the comfort of their homes and send detailed reports to their physicians. A prescription by a licensed physician is required for purchase of the PCA 500.
Click here for a demonstration video of the PCA 500: https://youtu.be/s96SmZrgXhE
A downloadable brochure for the PCA500 can be found at: https://dta0yqvfnusiq.cloudfront.net/usbst28863394/2019/02/PCA-500-ECG-system-5c59b033a49cd.pdf
ABOUT QT Medical
QT Medical, a visionary startup based outside of Los Angeles, California, has developed an at-home 12-lead ECG monitoring system that allows patients to record their own ECGs and share those tracings with their cardiologists. The FDA cleared Personal Cardiac Assistant (PCA 500) system provides hospital quality ECG recordings which can then be easily transferred to the patient’s smartphone using an accompanying app, and the data shared with the patient’s physician over QT Medical’s cloud platform. www.qtmedical.com
ABOUT MobilDrTech
MobilDrTech, Inc. is a full-service telemedicine systems and equipment provider. Founded in 2009 and based in Sugar Land, TX, MobilDrTech provides telemedicine services such as HIPAA compliant video conference platforms and functions as a Value-Added Reseller (VAR) for telemedicine equipment manufacturers including leading manufacturers of telemedicine stethoscopes, otoscopes, ECGs, general examination cameras and telemedicine carts. www.mobildrtech.com
MobilDrTech Releases White Paper on "Telemedicine Stethoscopes"
MobilDrTech, Inc. releases a white paper titled “Telemedicine Stethoscopes” that identifies and compares the most commonly used real time telemedicine stethoscopes in the U.S. market.
Sugar Land, TX, January 8, 2019—(PR.Com)—// Telehealth, Telemedicine, Stethoscopes, Healthcare, Industry News, Market News, Press Releases, Products and Services
MobilDrTech, a respected vendor of telemedicine equipment and diagnostic devices (www.mobildrtech.com) has released a white paper identifying and comparing design, features and cost of today’s most frequently utilized telemedicine stethoscopes.
Many healthcare providers are rapidly adding remote auscultation to their telehealth and telemedicine programs. This white paper is provided as a free resource to assist them in their product research and selection process.
The white paper explores the evolution of stethoscopes used specifically for telemedicine encounters. The history of telemedicine stethoscopes can be traced back almost 25 years to a stethoscope with an analog output that worked directly with normal telephone lines. From that point until today, development of newer and better hardware and software has continued unabated.
Three charts summarizing the features and cost comparisons from the document can be downloaded here: https://dta0yqvfnusiq.cloudfront.net/usbst28863394/2018/12/Telemedicine-Stethoscope-Charts-5c1fb7177dd61.pdf
Full text of the white paper can be downloaded here: https://dta0yqvfnusiq.cloudfront.net/usbst28863394/2019/01/Telemedicine-Stethoscopes-White-Paper-MDrT-1-8-2019-5c2ea3d3e4fd3.pdf
Also see: www.usbsteth.com
Chrome Compatible Telemedicine Stethoscope API in Beta
July 2, 2018 // Healthcare, Industry News, Market News, planetWebRTC.com, Press Releases, Products & Services, Telemedicine, WebRTC
VIERA, FL – JULY 02, 2018 – RNK Products, Inc. (RNK) has created a Chrome compatible “application programmers interface” (API) for its PCP-SSP stethoscope signal processing software. This new API will enable Value Added Resellers (VARs) to easily integrate the FDA cleared stethoscope signal processing software into their own platform.
Key features of the PCP-SSP/API software include:
- Capability to use the audio channel of the video conferencing to pass the stethoscope signal.
- User selectable audio filters to enable clinicians to focus on lung sounds and heart sounds separately.
- Local loop back to enable a clinician with the patient to listen to the same sounds as the remote clinician.
Because it is built on Chrome and well matched to WebRTC video conferencing, PCP-SSP/API will work on any platform that hosts Chrome.
The PCP-SSP/API software works with the high performance PCP-USB stethoscope that dominates hospital/clinic telemedicine applications, including in the VA network. The PCP-USB is also the easiest to use telemedicine stethoscope on the market. It is a plug-n-play device that plugs into any USB port. It needs no batteries and there are no patient controls.
“The trend in telemedicine system development is to browser based technology using WebRTC for the video conferencing and Chrome for the browser,” says Rich Abbruscato, CEO of RNK. “PCP-SSP/API software and the PCP-USB stethoscope are ideal for those telemedicine systems serving all levels of acuity.”
To meet the demand for real-time stethoscopes in browser based telemedicine systems, RNK is pleased to move to the Beta stage for its Chrome compatible PCP-SSP/API software.
More at http://www.rnkproducts.com.
Release of White Paper: Integrating Telemedicine Into EHRs
With telemedicine continuing to be at the forefront of the media, EHRGuide.org partnered with C. R. (Rich) Abbruscato, to author a white paper titled, "Integrating Telemedicine into EHRs."
May 30, 2018 --(PR.com)-- EHRGuide.org, a free online EHR resource for small to mid-sized medical practices, recently released a white paper exploring the integration of telemedicine into EHRs.
Besides helping break down the top EHR solutions to narrow down the choices, EHRGuide.org also provides industry insight on the hottest topics in the health information space.
With telemedicine continuing to be at the forefront of the media, EHRGuide.org partnered with C. R. (Rich) Abbruscato, to author a white paper titled, "Integrating Telemedicine into EHRs." With over 20 years in telemedicine, Rich is one of the pioneers in the telemedicine market. He is founder of RNK Products (using the brand Telehealth Technologies) a company dedicated to the design, development and manufacture of telemedicine medical devices (most notably real-time stethoscopes) and telemedicine systems.
This white paper is a composition of a four part series of articles that provides insight into just why so many organizations are struggling to integrate telemedicine into EHRs. To view the white paper in full, visit https://ehrguide.org/white-paper-integrating-telemedicine-into-ehrs/.
In this white paper, Rich helps the reader better understand the question that integrators ask themselves of “which EHR”? Then he goes on to explain “what not to do.” Learn about a sensible approach for a telemedicine-to-EHR interface and a possible breakthrough that the telehealth and EHR industry has been looking for.
More at https://ehrguide.org
MobilDrTech Announces Opening of Online Store with Posted Pricing and Popular Telemedicine Equipment
April 20, 2017 - MobilDrTech is pleased to announce the launch of an online store offering proven, mainstream telemedicine products that work well together at fixed, reasonable prices. www.telemedicinesupply.com
Are you starting your first telemedicine clinic, opening an additional one or just adding devices to your existing program?
MobilDrTech has put together an excellent set of products and, unlike larger telemedicine resellers, does not try to sell everything that anyone could possibly need. Neither do we limit our product line to a single manufacturer or try to lock you into certain product lines. We provide the best mainstream telemedicine products we have found and used successfully with our customers in a wide range of telemedicine settings.
In the Online Store, MobilDrTech offers:
- HIPAA Compliant Video Conference Platforms
- Otoscopes
- Stethoscopes
- General Examination Cameras
- Mobile Telemedicine Carts
- Portable Integrated Cases
- Do It Yourself Telemedicine Kits
- Accessories
“Our goal is to operate a store that offers providers what they really need quickly, efficiently and at a reasonable cost. If we do that, we will be very successful.” says Robert Jenkins, Director of Customer Services for MobilDrTech.
MobilDrTech will be featuring the telemedicinesupply.com website at Booth 2040 at the American Telemedicine Association conference in Orlando April 23 – 26. Please stop by for more information or check us out online at www.telemedicinesupply.com .
About MobilDrTech:
MobilDrTech, Inc. is a full-service telemedicine equipment and software provider based in Sugar Land, TX. The company provides HIPAA compliant video conference platforms and is a Value Added Reseller (VAR) for leading manufacturers of telemedicine stethoscopes, otoscopes, dermatoscopes, general examination cameras and fully integrated telemedicine carts, cases and DIY Kits.
www.mobildrtech.com
Press Release: RNK Releases FDA Class II Cleared Telemedicine Stethoscope Software
July 7, 2016 - RNK is pleased to announce the release of a simplified version of its FDA approved sSOIP telemedicine stethoscope software. This new software version is called PCP-SSP and works with RNK’s flagship PCP-USB stethoscope.
Key PCP-SSP features:
- Enables the stethoscope signal to be sent over the audio channel of a video conferencing connection. No separate network connection needed.
- Can automatically mute the room audio when stethoscope sounds are being sent.
- Provides filtering options for the clinicians to focus on heart sounds and lung sounds separately.
- Provides a local monitor capability so that the clinician with the patient can hear the same stethoscope sounds as the remote clinician.
Remote auscultation involves two software elements on the PC: 1) stethoscope signal processing and 2) network transport. PCP-SSP lets the video conferencing equipment handle the network connectivity as an MDDS, while it handles the stethoscope signal processing as an FDA cleared medical device. By performing the stethoscope signal processing functions and satisfying FDA requirements, PCP-USB relieves the video conferencing system of both technical and regulatory responsibilities.
All electronic stethoscopes are not approved for telemedicine. For example, an electronic stethoscope with an analog connection to the Microphone port of the telemedicine system PC requires that the PC perform the analog-to-digital conversion (ADC) and digital encoding. By doing that such a telemedicine system has inadvertently become subject to FDA regulations for a Class II medical device.
“We are very pleased to be able to respond to customer demand for a straight forward telemedicine stethoscope package that works with any Windows PC based video conferencing system and meets key clinical user needs while maintaining FDA compliance” says Rich Abbruscato, CEO of RNK.
About RNK Products, Inc.
Founded in 2003, RNK Products, Inc. specializes in electronic and telephonic stethoscopes, under both its own label Telehealth Technologies, and under private labels used by its Value-Added Resellers. RNK offers a range of stethoscopes to fit any institutional or home telemedicine program.